Ultrasonic Solubilization of Protein Pellets
In proteomics, sample preparation is never a minor detail. It is the foundation on which identification accuracy, quantification reliability, and reproducibility are built. One of the most persistent challenges in protein sample preparation is the efficient re-dissolution of protein pellets after precipitation or concentration steps. This is where ultrasonic solubilization of protein pellets has become increasingly important. By applying controlled sonication, laboratories can improve protein recovery, accelerate pellet dissolution, and prepare samples more effectively for downstream mass spectrometry and biochemical analysis.
Protein Solubilization: Why Sonication Matters in Modern Proteomics
Protein pellets often form during acetone, ethanol, methanol-chloroform, ammonium sulfate, or TCA precipitation. These workflows are widely used to remove contaminants, concentrate proteins, and purify extracts before analysis. Yet once precipitation is complete, the resulting pellet can be difficult to re-solubilize. Dense aggregates, hydrophobic domains, membrane-associated proteins, and strongly interacting protein complexes frequently resist conventional mixing or vortexing. Incomplete solubilization can then lead to sample loss, biased representation of proteins, and poor reproducibility across experiments.
Sonication addresses exactly this bottleneck. Through the generation of mechanical energy in a liquid medium, sonication disrupts compact pellet structures, promotes buffer penetration, and disperses aggregated material into solution. The result is a faster and often more complete reconstitution of proteins, which is particularly valuable when working with limited samples, complex lysates, or challenging proteomic targets.

Microplate Sonicator UIP400MTP for protein extraction and pellet solubilization
Why Protein Pellets Are Difficult to Solubilize
 Protein precipitation is effective because it forces proteins out of solution. However, the same process that makes precipitation useful also creates the problem of pellet recovery. Once pelleted, proteins may become tightly packed and partially denatured. Hydrophobic interactions can intensify, intermolecular binding can increase, and some proteins may trap salts, lipids, nucleic acids, or other matrix components. Even when a strong solubilization buffer is used, passive resuspension is often slow and incomplete.
Protein precipitation is effective because it forces proteins out of solution. However, the same process that makes precipitation useful also creates the problem of pellet recovery. Once pelleted, proteins may become tightly packed and partially denatured. Hydrophobic interactions can intensify, intermolecular binding can increase, and some proteins may trap salts, lipids, nucleic acids, or other matrix components. Even when a strong solubilization buffer is used, passive resuspension is often slow and incomplete.
In proteomics, this matters because incomplete pellet dissolution does not just reduce total yield. It can selectively exclude certain protein classes, especially membrane proteins, structural proteins, or aggregation-prone species. That means the final analytical result may no longer reflect the true composition of the original sample. In high-resolution proteomics, where subtle differences in abundance or post-translational modification can be biologically decisive, such preparation bias is a serious limitation.
How Sonication Improves Protein Pellet Solubilization
Ultrasonic treatment improves solubilization by introducing high-frequency mechanical energy into the sample. This energy helps break apart compact pellet material and increases the contact between the solubilization buffer and the embedded proteins. Instead of relying only on diffusion and manual mixing, the process actively disperses the pellet into smaller fractions that are easier to dissolve.
The practical effect is significant. Sonication can:
- accelerate dissolution of dense or stubborn protein pellets
- improve recovery of poorly soluble and aggregated proteins
- reduce preparation time in proteomics workflows
- support more homogeneous samples for digestion and analysis
This enhanced dispersion is especially useful when pellets are resuspended in buffers containing urea, thiourea, detergents, chaotropes, or other reagents commonly used in proteomics. Sonication helps these components reach and solubilize the pellet more efficiently, resulting in a more uniform sample solution.
Advantages of Ultrasonic Solubilization in Proteomics
The key advantage of ultrasonic solubilization is that it turns a frequently underestimated preparation step into a controllable and efficient process. In proteomics, that has direct analytical consequences.
- First, improved solubilization increases the likelihood that the sample entering enzymatic digestion is representative of the full protein population. Trypsin digestion, for example, depends on proteins being adequately unfolded and accessible in solution. If parts of the pellet remain undissolved, those proteins are effectively excluded from peptide generation and therefore from detection.
- Second, sonication can improve reproducibility. Manual pellet resuspension is inherently variable, especially when different operators, pellet sizes, or sample matrices are involved. Controlled ultrasonic treatment standardizes the physical energy applied to the sample, which can reduce variability between preparations and improve consistency in downstream LC-MS or gel-based workflows.
- Third, ultrasonication is highly valuable for low-input and precious samples. Clinical proteomics, biomarker discovery, cell culture experiments, and tissue studies often rely on limited material. Any protein loss during solubilization reduces the informational value of the sample. Efficient ultrasonic re-dissolution helps preserve as much analyte as possible.
- Finally, sonication supports workflow speed. Proteomics laboratories processing multiple samples need robust, time-efficient preparation methods. A pellet that dissolves quickly and completely is not just convenient; it reduces delays, lowers the risk of handling errors, and improves throughput.
Sonication vs Conventional Resuspension Methods
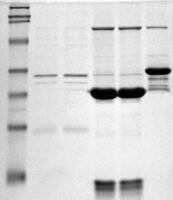 Traditional pellet resuspension methods usually involve pipetting, stirring, vortexing, prolonged incubation, or repeated heating steps. While these techniques can work for loosely packed pellets, they often struggle with highly compact or hydrophobic protein material. Mechanical mixing alone may fail to disintegrate the pellet structure fully, leaving visible particulates or invisible insoluble fractions behind.
Traditional pellet resuspension methods usually involve pipetting, stirring, vortexing, prolonged incubation, or repeated heating steps. While these techniques can work for loosely packed pellets, they often struggle with highly compact or hydrophobic protein material. Mechanical mixing alone may fail to disintegrate the pellet structure fully, leaving visible particulates or invisible insoluble fractions behind.
Sonication provides a more active and targeted approach. Rather than depending on slow buffer diffusion, it physically disrupts the pellet and promotes rapid homogenization. This does not eliminate the need for an appropriate resuspension buffer, but it substantially enhances the performance of that buffer.
Compared with purely manual methods, ultrasonic solubilization often offers better process control, greater efficiency, and improved suitability for demanding proteomics applications. For laboratories seeking both analytical quality and operational reliability, this makes sonication a compelling choice.
Best Use Cases for Ultrasonic Protein Pellet Solubilization
Ultrasonic solubilization is especially beneficial in workflows involving:
- protein precipitation before mass spectrometry,
- reconstitution of pellets from cell lysates or tissue extracts,
- recovery of membrane-rich or aggregation-prone proteins,
- and sample preparation for quantitative proteomics where reproducibility is essential.
It is also highly relevant when pellets have been stored, dried too strongly, or produced from complex biological matrices. In such cases, passive resuspension can become particularly inefficient, while sonication helps restore sample usability with less manual intervention.

VialTweeter sonicator for the simultaneous sonication of 10 samples, e.g. for protein extraction and solubilization
Find the Best Sonicator for Your Protein Solubilization Workflow!
For laboratories working with precious samples, low-input material, or high-throughput proteomics, Hielscher’s portfolio offers several sonication formats that can be matched precisely to the workflow.
Wether you choose a Hielscher probe-type sonicator, the VialTweeter Multi-Tube Sonicator, or the UIP400MTP Microplate Sonicator – each ultrasonicator model addresses a different sample preparation scenario while sharing the same core advantage: reproducible ultrasonic energy for efficient and controlled sample processing.
Probe-Type Sonicators
 Ultrasonic probes such as the UP200Ht are particularly well suited for direct sonication of individual samples. For proteomics labs, the UP200Ht is a strong choice when protein pellets need intensive resuspension in small to medium volumes, especially where method control and repeatability are important. Direct probe sonication can rapidly disrupt compact pellet material and help solubilization buffers access proteins that would otherwise remain partially undissolved.
Ultrasonic probes such as the UP200Ht are particularly well suited for direct sonication of individual samples. For proteomics labs, the UP200Ht is a strong choice when protein pellets need intensive resuspension in small to medium volumes, especially where method control and repeatability are important. Direct probe sonication can rapidly disrupt compact pellet material and help solubilization buffers access proteins that would otherwise remain partially undissolved.
Overview of all probe-type sonicators!
VialTweeter Multi-Tube Sonicator
 Where multiple closed vials must be processed under identical conditions, the Multi-Tube Sonicator VialTweeter offers a distinct advantage. The VialTweeter allows for intensive sonication of small volumes sonicating several closed vials under sterile conditions. The simultaneous sample preparation in multiple test tubes under the same conditions, as well as the reduced risk of cross-contamination, sample loss, and aerosol formation during closed-vial processing make the VialTweeter a reliable tool for sample preparation. For proteomics, this is highly relevant when handling valuable pellets from multiple replicates or clinical samples, where consistency between tubes is critical.
Where multiple closed vials must be processed under identical conditions, the Multi-Tube Sonicator VialTweeter offers a distinct advantage. The VialTweeter allows for intensive sonication of small volumes sonicating several closed vials under sterile conditions. The simultaneous sample preparation in multiple test tubes under the same conditions, as well as the reduced risk of cross-contamination, sample loss, and aerosol formation during closed-vial processing make the VialTweeter a reliable tool for sample preparation. For proteomics, this is highly relevant when handling valuable pellets from multiple replicates or clinical samples, where consistency between tubes is critical.
Learn more about the VialTweeter!
Microplate Sonicator UIP400MTP
 For high-throughput laboratories, the UIP400MTP microplate sonicator extends the benefits of sonication into plate-based workflows. The UIP400MTP as a microplate and multi-well plate sonicator for uniform ultrasonic processing across standard plates, including 96-well formats, and emphasizes its suitability for automated sample preparation in fields such as proteomics, diagnostics, and drug discovery. The platform is designed for simultaneous treatment of many samples, with advantages such as reduced cross-contamination risk, lower labor intensity, improved sample recovery, and integration into automated workflows.
For high-throughput laboratories, the UIP400MTP microplate sonicator extends the benefits of sonication into plate-based workflows. The UIP400MTP as a microplate and multi-well plate sonicator for uniform ultrasonic processing across standard plates, including 96-well formats, and emphasizes its suitability for automated sample preparation in fields such as proteomics, diagnostics, and drug discovery. The platform is designed for simultaneous treatment of many samples, with advantages such as reduced cross-contamination risk, lower labor intensity, improved sample recovery, and integration into automated workflows.
In practical proteomics, this means that pellet solubilization, cell lysis, extraction, and related preparation steps can be scaled much more efficiently. Rather than processing samples one by one, labs can sonicate entire plates with consistent energy input. This is valuable whenever workflows need to combine throughput with analytical rigor, for example in screening studies, quantitative proteomics, or standardized sample preparation pipelines. The UIP400MTP is therefore not just a convenience tool; it is a platform that supports the broader trend toward automation, reproducibility, and robust high-throughput proteomics.
Learn more about the UIP400MTP Microplate Sonicator!

High-throughput protein extraction and solubilization with the microplate sonicator UIP400MTP
Design, Manufacturing and Consulting – Quality Made in Germany
Hielscher ultrasonicators are well-known for their highest quality and design standards. Robustness and easy operation allow the smooth integration of our ultrasonicators into industrial facilities. Rough conditions and demanding environments are easily handled by Hielscher ultrasonicators.
Hielscher Ultrasonics is an ISO certified company and put special emphasis on high-performance ultrasonicators featuring state-of-the-art technology and user-friendliness. Of course, Hielscher ultrasonicators are CE compliant and meet the requirements of UL, CSA and RoHs.
Literature / References
- FactSheet UIP400MTP Plate-Sonicator for High-Throughput Sample Preparation – English version – Hielscher Ultrasonics
- FactSheet VialTweeter – Sonicator for Simultaneous Sample Preparation
- Susana Jorge, Kevin Pereira, Hugo López-Fernández, William LaFramboise, Rajiv Dhir, Javier Fernández-Lodeiro, Carlos Lodeiro, Hugo M. Santos, Jose L. Capelo-Martínez (2020): Ultrasonic-assisted extraction and digestion of proteins from solid biopsies followed by peptide sequential extraction hyphenated to MALDI-based profiling holds the promise of distinguishing renal oncocytoma from chromophobe renal cell carcinoma. Talanta, Volume 206, 2020.
- Lindemann C, Lupilova N, Müller A, Warscheid B, Meyer HE, Kuhlmann K, Eisenacher M, Leichert LI. (2013): Redox proteomics uncovers peroxynitrite-sensitive proteins that help Escherichia coli to overcome nitrosative stress. Journal of Biological Chemistry 288(27); 2013. 19698-714.
- Gonçalo Martins, Javier Fernández-Lodeiro, Jamila Djafari, Carlos Lodeiro, J.L. Capelo, Hugo M. Santos (2019): Label-free protein quantification after ultrafast digestion of complex proteomes using ultrasonic energy and immobilized-trypsin magnetic nanoparticles. Talanta, Volume 196, 2019. 262-270.
Frequently Asked Questions
Why are Ultrasonic Baths Not Suitable for Protein Solubilization?
In ultrasonic baths, cavitation, the working principle of sonication, occurs very uneven disposing samples to varying sonication treatments. Depending on the position of sample tubes in the ultrasonic bath, each sample is impacted by different intensities. Proteomics depends on comparability. If one pellet is incompletely dissolved while another is fully resuspended, the resulting data may reflect preparation bias rather than true biology. In contrast to ultrasonic baths, non-contact sonicators such as the VialTweeter or the Microplate Sonicator UIP400MTP support more standardized handling by enabling several samples to be processed in parallel under matching ultrasonic conditions, which can help improve reproducibility across experiments. This is particularly useful in biomarker studies, comparative proteomics, and workflows with multiple biological or technical replicates.
What are the Most Common Assays in Proteomics?
The most common assays in proteomics are protein quantification assays and protein characterization methods used during sample preparation and analysis. Frequently used assays include the Bradford assay, BCA assay, Lowry assay, and UV absorbance at 280 nm for protein concentration measurement. In broader proteomics workflows, SDS-PAGE, Western blotting, ELISA, in-gel digestion, and mass spectrometry-based analyses are also widely used to assess protein abundance, purity, molecular weight, and identity.
What is Coomassie Brilliant Blue?
Coomassie Brilliant Blue is a triphenylmethane dye widely used in protein science for staining proteins in gels and for colorimetric protein quantification. It binds primarily to basic and aromatic amino acid residues, especially arginine, and undergoes a spectral shift upon binding to proteins. This property makes it useful both for visualizing proteins after electrophoresis and for the Bradford protein assay.
How does the Bradford Assay Work?
The Bradford assay works by mixing a protein sample with Coomassie Brilliant Blue dye under acidic conditions. When the dye binds to proteins, its absorbance maximum shifts from about 465 nm to 595 nm, causing a measurable color change from reddish-brown to blue. The increase in absorbance at 595 nm is proportional to the protein concentration over a defined range, allowing quantification by comparison with a standard curve, usually prepared with bovine serum albumin.

Hielscher Ultrasonics manufactures high-performance ultrasonic homogenizers from lab to industrial size.



