Ultrasonic Liposome Encapsulation of GLP-1 Peptides: Scalable Technology for Oral Peptide Delivery
Semaglutide and other GLP-1 peptides are transforming diabetes and obesity care, but most peptide drugs still require injections because oral delivery remains extremely challenging. Even approved oral semaglutide shows bioavailability below 1%, increasing dose requirements and cost. Encapsulating GLP-1 peptides in liposomes can overcomes these limitations. Learn how ultrasonic liposome preparation facilitates GLP-1 peptide-loaded liposomes in compounding pharmacies and pharmaceutical manufacturing.
Liposomal GLP-1 Peptides
GLP-1 receptor agonists such as semaglutide and next-generation incretin peptides such as tirzepatide have transformed the treatment of type 2 diabetes and obesity. Yet, from a formulation and manufacturing perspective, these APIs still come with the classic limitation of peptide drugs: they are fragile, easily degraded, and difficult to deliver through the gastrointestinal tract.
That is why most GLP-1 peptides remain injectable, even though oral delivery would dramatically reduce barriers for patients. Oral delivery generally improves convenience, adherence, and willingness to start therapy earlier – especially in chronic diseases.
However, peptide oral delivery remains one of the most difficult challenges in pharmaceutical science. Even approved oral semaglutide products show very low bioavailability, requiring high doses and careful formulation strategies.
One of the most promising technical approaches to overcome these limitations is liposomal encapsulation, combined with high-intensity ultrasonic processing. Liposomes protect peptide APIs, improve dispersion stability, and can be engineered for nano-scale size ranges that are relevant for mucus penetration and intestinal interaction. Sonication provides a scalable and reproducible method to produce these liposomes at industrially relevant volumes.
Why Liposomes Are a Strong Platform for GLP-1 Peptides
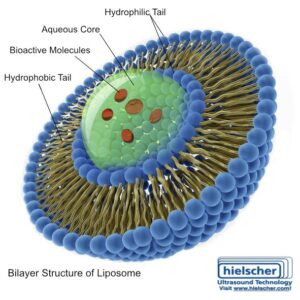 Liposomes are vesicles formed by phospholipid bilayers, similar in composition to biological membranes. This makes them inherently biocompatible and well suited for pharmaceutical development. For peptide drugs, liposomes are attractive because they can physically protect the API and provide a formulation architecture that can be tuned for delivery performance.
Liposomes are vesicles formed by phospholipid bilayers, similar in composition to biological membranes. This makes them inherently biocompatible and well suited for pharmaceutical development. For peptide drugs, liposomes are attractive because they can physically protect the API and provide a formulation architecture that can be tuned for delivery performance.
In the context of GLP-1 peptides, the key challenge is not only protecting the peptide from acid and enzymes, but also enabling meaningful uptake across the intestinal barrier. This is a multi-layer problem involving pH, enzymatic degradation, mucus transport, epithelial permeability, and transport mechanisms.
The scientific literature increasingly supports the idea that vesicle-based carriers can improve peptide stability and interaction with intestinal systems.
Why Semaglutide Is Particularly Interesting for Liposomal Encapsulation
Semaglutide is not just a peptide – it is a peptide with a built-in lipid tail modification. This is a key reason it behaves differently from many other peptide APIs in lipid systems.
GLP-1 peptides such as semaglutide and tirzepatide load into vesicles via lipid-tail insertion into the vesicle membrane.
From an engineering perspective, this is important because it suggests semaglutide is structurally compatible with lipid bilayers, meaning it may be possible to achieve stable association and meaningful loading without excessive chemical modification.
This same logic applies to other lipidated peptide APIs and peptide-drug conjugates.
The Manufacturing Reality: Liposomes Must Be Reproducible and Scalable
Liposome encapsulation is not difficult in a lab. The true difficulty begins when a formulation must be manufactured in a reproducible way at scale.
Industrial liposome production must control:
- liposome size distribution
- polydispersity
- encapsulation efficiency
- dispersion stability
- batch-to-batch reproducibility
- sterile processing compatibility
- process validation and documentation
Many common liposome methods (vortexing, simple hydration, hand extrusion) can produce good results at the bench – but they fail when moved to pilot or production volumes.
This is where ultrasonic processing becomes a key enabling technology.
Ultrasonic Liposome Encapsulation: The Core Principle
High-intensity ultrasound introduces mechanical energy into a liquid via acoustic cavitation. Cavitation is the formation and collapse of microscopic bubbles, producing localized shear forces and micro-mixing effects.
In lipid dispersions, these forces:
- create micro-emulsions to initiate liposome formation
- break down large lipid aggregates
- reduce multilamellar structures
- generate smaller, more uniform vesicles
- improve homogenization
- increase the reproducibility of the final dispersion
Ultrasonic processing is therefore widely used in nanomaterial production, emulsification, and dispersion – and it is highly suited for liposome nano-sizing.
For GLP-1 peptide liposomes, ultrasound can be used either to create liposomes directly during hydration and dispersion, or as a post-processing step to refine vesicle size and improve uniformity.
Why Ultrasound Is Especially Valuable for Pharmaceutical Liposome Manufacturing
The single most important reason ultrasound is used industrially is that it can be scaled by controlling a measurable process parameter: energy per volume.
Instead of scaling by “more mixing” or “longer processing,” ultrasonic systems allow the process to be scaled by:
- increasing ultrasonic power
- increasing flow rate
- maintaining the same energy input per mL
- using continuous flow reactors
- numbering-up in parallel
This makes the process highly transferable from R&D to production.
In practical terms, this means a liposome process developed on a small system can be moved to larger systems while maintaining equivalent process conditions, which is exactly what pharmaceutical manufacturing requires.
The Impact on GLP-1 Peptides: Toward Better Oral Bioavailability
Oral delivery is the long-term objective for many GLP-1 peptide formulations. The reason is straightforward: if oral bioavailability improves, the entire therapy becomes easier for patients.
Your uploaded material highlights the current limitation: oral semaglutide is possible, but bioavailability remains low (below 1%).
Liposome encapsulation is not a guarantee of high oral bioavailability, but it addresses several critical bottlenecks simultaneously:
- It can physically protect the peptide against degradation.
- It can create nanoscale carriers with properties tuned for mucus penetration.
- It can be functionalized with surface ligands for active transport mechanisms.
- It can improve consistency and reproducibility of the delivered API form.
For semaglutide in particular, membrane association via the lipid tail provides an additional mechanism that may stabilize the peptide in lipid systems.
Ultrasonic Processing Enables Continuous, Industrial Liposome Production
In industrial manufacturing, continuous processing is often preferred because it improves:
- throughput
- reproducibility
- process control
- integration into sterile production workflows
Ultrasonic flow-through systems are ideal for this. The liposome dispersion is pumped through a pressurized flow cell where ultrasound is applied under controlled conditions. Temperature, pressure, and residence time can be controlled, which is essential for peptide formulations.
This enables scalable nano-sizing and encapsulation workflows that are far closer to GMP requirements than many laboratory-only methods.
Hielscher Ultrasonics: Lab-to-Industrial Systems for Liposome Encapsulation

Hielscher Ultrasonics provides ultrasonic systems that match the real development pathway of liposomal peptide formulations.
At the laboratory scale, the UP400St is widely used for formulation screening, process development, and compounding-scale liposome preparation.
At the manufacturing scale, the UIP2000hdT and UIP4000hdT are designed for industrial duty cycles and can be integrated with flow-through sonication cells for continuous processing.
This combination is particularly relevant for pharmaceutical liposome production because it supports:
- process reproducibility
- linear scalability
- continuous flow manufacturing
- high-power ultrasonic processing under controlled conditions

Sonicator UIP1000hdT with glass flow cell for the production of liposomes.
At a Glance – Why Ultrasonic Liposome Formulation of GLP-1 Peptides
Ultrasonic liposome encapsulation is one of the most technically mature and industrially scalable approaches for producing peptide-loaded liposomes. For GLP-1 peptides such as semaglutide and tirzepatide, this approach is especially relevant because these APIs are structurally compatible with lipid membranes and can benefit from vesicle-based protection and delivery strategies.
Most importantly, ultrasound is not merely a laboratory method – it is a scalable process technology. By controlling energy input per volume and using flow-through sonication cells, ultrasonic processing can be transferred from lab-scale development to pilot and full industrial production.
As the pharmaceutical industry continues moving toward peptide therapeutics – and as demand for patient-friendly delivery routes grows – ultrasonic liposome manufacturing will play an increasingly central role in enabling the next generation of GLP-1 formulations.
The table below gives you an indication of the approximate processing capacity of our ultrasonicators:
| Batch Volume | Flow Rate | Recommended Devices |
|---|---|---|
| 0.5 to 1.5mL | n.a. | VialTweeter |
| 1 to 500mL | 10 to 200mL/min | UP100H |
| 10 to 2000mL | 20 to 400mL/min | UP200Ht, UP400St |
| 0.1 to 20L | 0.2 to 4L/min | UIP2000hdT |
| 10 to 100L | 2 to 10L/min | UIP4000hdT |
| 15 to 150L | 3 to 15L/min | UIP6000hdT |
| n.a. | 10 to 100L/min | UIP16000hdT |
| n.a. | larger | cluster of UIP16000hdT |
Design, Manufacturing and Consulting – Quality Made in Germany
Hielscher ultrasonicators are well-known for their highest quality and design standards. Robustness and easy operation allow the smooth integration of our ultrasonicators into industrial facilities. Rough conditions and demanding environments are easily handled by Hielscher ultrasonicators.
Hielscher Ultrasonics is an ISO certified company and put special emphasis on high-performance ultrasonicators featuring state-of-the-art technology and user-friendliness. Of course, Hielscher ultrasonicators are CE compliant and meet the requirements of UL, CSA and RoHs.
Literature / References
- Martyna Truszkowska; Ahmad Saleh; Melanie Lena Eber; Gergely Kali; Andreas Bernkop-Schnürch (2025): Addressing the polycation dilemma in drug delivery: charge-converting liposomes. Journal of Materials Chemistry B 2025, 13, 9100-9111.
- Pop, R.; Nistor, M.; Socaciu, C.; Cenariu, M.; Tăbăran, F.; Rugină, D.; Pintea, A.; Socaciu, M.A. (2025): Distinct In Vitro Effects of Liposomal and Nanostructured Lipid Nanoformulations with Entrapped Acidic and Neutral Doxorubicin on B16-F10 Melanoma and Walker 256 Carcinoma Cells. Pharmaceutics 2025, 17, 904.
- M.E. Barbinta-Patrascu, N. Badea, M. Constantin, C. Ungureanu, C. Nichita, S.M. Iordache, A. Vlad, S. Antohe (2018): Bio-Activity of Organic/Inorganic Photo-Generated Composites in Bio-Inspired Systems. Romanian Journal of Physics 63, 702 (2018).
- Raquel Martínez-González, Joan Estelrich, Maria Antònia Busquets (2016): Liposomes Loaded with Hydrophobic Iron Oxide Nanoparticles: Suitable T2 Contrast Agents for MRI. International Journal of Molecular Science 2016.
- Shah Purvin, Parameswara Rao Vuddanda, Sanjay Kumar Singh, Achint Jain, and Sanjay Singh (2014): Pharmacokinetic and Tissue Distribution Study of Solid Lipid Nanoparticles of Zidov in Rats. Journal of Nanotechnology, Volume 2014.
- Harshita Krishnatreyya, Sanjay Dey, Paulami Pal, Pranab Jyoti Das, Vipin Kumar Sharma, Bhaskar Mazumder (2019): Piroxicam Loaded Solid Lipid Nanoparticles (SLNs): Potential for Topical Delivery. Indian Journal of Pharmaceutical Education and Research Vol 53, Issue 2, 2019. 82-92.
Frequently Asked Questions
What Role plays Ultrasonic Emulsification in Liposome Formation?
Ultrasonic emulsification of the aqueous phase and lipids provides the mechanical energy required to disperse lipids finely in water and to drive their self-assembly into closed bilayer structures. Acoustic cavitation generated by ultrasound creates intense micro-mixing and shear forces that fragment lipid phases, hydrate lipid molecules uniformly, and convert dispersed lipid fragments into liposomal vesicles. This promotes rapid vesicle formation, reduces multilamellar structures, and yields smaller, more uniform liposomes with improved reproducibility and stability.
What Should I Know about GLP-1 Formulating?
GLP-1 peptides are highly effective drugs, but most remain injectable due to GI degradation and absorption barriers.
Oral semaglutide exists, but reported bioavailability remains below 1%.
Semaglutide and tirzepatide can be loaded into vesicle systems, and loading efficiency depends strongly on the processing method.
Semaglutide may associate with lipid membranes via its lipid tail, supporting vesicle/liposome compatibility.
Ultrasonic cavitation enables reproducible liposome nano-sizing and dispersion homogenization.
Ultrasound scales linearly via energy-per-volume control and continuous flow processing.
Hielscher systems support the full workflow:
- UP400St (lab and compounding scale)
- UIP2000hdT / UIP4000hdT + flow cells (industrial pharmaceutical manufacturing)
What are GLP-1 Peptides?
GLP-1 peptides are peptide-based incretin receptor agonists that mimic or enhance the biological activity of glucagon-like peptide-1 (GLP-1), a gut hormone involved in glucose-dependent insulin secretion, suppression of glucagon release, and regulation of gastric emptying and appetite. Clinically used GLP-1 drugs (e.g., semaglutide) are chemically modified to resist enzymatic degradation and achieve long systemic half-life.
How are GLP-1 Peptides Commonly Administered?
Most GLP-1 peptides are administered by subcutaneous injection because peptides are unstable in the gastrointestinal tract and have extremely low intestinal permeability. Oral administration has been achieved for semaglutide using specialized formulation strategies, but oral bioavailability remains low (reported around 0.4%–1%).
What are the Advantages of Oral Administration of GLP-1 Peptide Liposomes?
Oral GLP-1 peptide liposomes can improve patient convenience and adherence by eliminating injections while potentially increasing effective absorption by protecting peptides from acid and enzymatic degradation and improving transport through mucus and epithelial barriers. Improved oral bioavailability can reduce the dose burden, lower cost pressure, and reduce barriers for patients to start and maintain long-term peptide therapy.

Hielscher Ultrasonics manufactures high-performance ultrasonic homogenizers from lab to industrial size.



