Liposomal Semaglutide Could Be the Next Big Leap in GLP-1 Drug Delivery
, Kathrin Hielscher, published in Hielscher News
GLP-1 peptides such as semaglutide has become one of the most influential peptide therapeutics of the last decade, with broad clinical use in type 2 diabetes and obesity. Yet despite its clinical success, the formulation and manufacturing challenges of semaglutide remain representative of peptide drugs in general: they are structurally fragile, difficult to protect against degradation, and notoriously hard to deliver by non-invasive routes. These limitations are a major reason why most GLP-1 receptor agonists still rely on injections, even as demand for oral or patient-friendly delivery continues to grow.
Limits of Current Oral GLP-1 Formulations
The development of oral GLP-1 peptides demonstrated that oral delivery is technically possible, but it also exposed the core limitations of existing strategies. Even in an approved product, oral semaglutide exhibits very low bioavailability, typically below 1%, which necessitates higher dosing and contributes to cost, variability, and formulation complexity. These constraints have intensified interest in carrier-based systems that can protect peptide drugs and potentially improve absorption without relying exclusively on chemical permeation enhancers.
Liposomes as a Technically Mature Delivery Platform
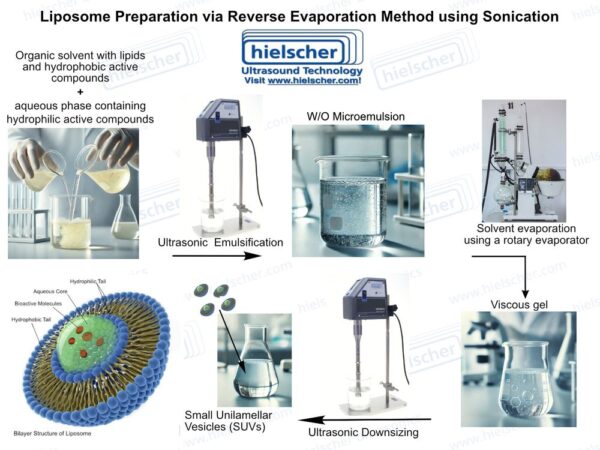
 Among the delivery systems under investigation, liposomal encapsulation stands out for its technical maturity and pharmaceutical relevance. Liposomes consist of phospholipid bilayers that closely resemble biological membranes and have a long history of clinical use in oncology and infectious disease. Their relevance for peptide therapeutics lies in their ability to physically protect sensitive APIs while offering tunable size, composition, and surface properties. However, liposomal performance is strongly dependent on size distribution, bilayer structure, loading strategy, and manufacturing reproducibility – factors that are governed primarily by process technology rather than formulation composition alone.
Among the delivery systems under investigation, liposomal encapsulation stands out for its technical maturity and pharmaceutical relevance. Liposomes consist of phospholipid bilayers that closely resemble biological membranes and have a long history of clinical use in oncology and infectious disease. Their relevance for peptide therapeutics lies in their ability to physically protect sensitive APIs while offering tunable size, composition, and surface properties. However, liposomal performance is strongly dependent on size distribution, bilayer structure, loading strategy, and manufacturing reproducibility – factors that are governed primarily by process technology rather than formulation composition alone.
Why Ultrasonic Processing Is Central to Liposome Manufacturing
Ultrasonic processing addresses several of the core challenges associated with liposome production. High-intensity ultrasound generates acoustic cavitation in liquids, producing localized shear forces and micro-mixing effects that can break down lipid aggregates and convert multilamellar structures into smaller, more uniform vesicles. In liposome manufacturing, ultrasound can be applied during vesicle formation or as a post-processing step to standardize particle size and dispersion quality. This dual role makes ultrasound particularly valuable in controlling critical quality attributes of liposomal systems.
Structural Compatibility of Semaglutide with Lipid Bilayers
GLP-1 peptides such as semaglutide or tirzepatide is especially well suited for lipid-based carriers because it is not a simple linear peptide. The molecule contains a chemically modified lipid tail that promotes interaction with lipid membranes. Experimental studies using vesicle systems have shown that semaglutide and related peptides can associate with vesicle membranes through insertion of this lipid tail. Although these findings were obtained using milk-derived extracellular vesicles rather than synthetic liposomes, the underlying mechanism is directly transferable. Lipidated peptides have an inherent affinity for phospholipid bilayers, which can improve loading efficiency and formulation stability without requiring complex chemical conjugation.
Process Conditions Determine Encapsulation Efficiency
A critical insight from recent vesicle-based studies is that encapsulation efficiency is highly dependent on the loading and processing method. This has important implications for pharmaceutical development: the success or failure of a peptide liposome formulation often depends less on the choice of lipid and more on how the vesicles are produced and processed. Ultrasonic processing provides a controllable and reproducible means of influencing these parameters, making it particularly attractive for systematic formulation development.
Scalability as a Key Advantage of Ultrasonic Processing
From a manufacturing standpoint, one of the most significant advantages of ultrasound is its scalability. Unlike many nanoparticle production techniques that rely on geometry-specific batch conditions, ultrasonic processing can be scaled by controlling energy input per unit volume. This allows processes developed at laboratory scale to be transferred to pilot and industrial systems with high comparability. For pharmaceutical manufacturers, this characteristic supports reproducibility, validation, and efficient technology transfer across development stages.
Continuous Flow Sonication for Industrial Production
The most industrially relevant implementation of ultrasonic liposome processing is continuous flow operation. In flow-through sonication cells, liposome dispersions pass through a defined reactor volume while ultrasound is applied under controlled pressure, amplitude, and temperature. This configuration enables precise control over residence time and energy exposure. For peptide-loaded liposomes, where thermal sensitivity and structural integrity are critical, such control is essential to maintaining product quality at scale.
Relevance for Next-Generation GLP-1 and Peptide Therapeutics
As GLP-1 therapies evolve toward dual- and multi-agonist peptides, formulation complexity is expected to increase. At the same time, patient demand for oral or less invasive delivery routes continues to grow. Scalable carrier-based delivery platforms therefore become strategically important, not only for improving pharmacokinetics but also for ensuring that new peptide drugs can be manufactured reliably at commercial scale.
Addressing the Fundamental Barrier of Oral Peptide Delivery
The gastrointestinal tract is inherently hostile to peptides, and low oral bioavailability remains a fundamental barrier even for advanced formulations. Liposomal encapsulation does not eliminate this challenge, but it provides a rational engineering approach to reduce degradation and control how peptides interact with intestinal environments. When combined with scalable processing technologies such as ultrasound, liposome-based delivery systems move closer to industrial feasibility rather than remaining confined to laboratory experimentation.
From Laboratory Development to Industrial Implementation
 In practical development workflows, Hielscher Ultrasonics systems are frequently used as reference platforms for ultrasonic liposome processing. At laboratory and formulation-development scale, compact ultrasonic probes such as the UP200Ht and UP400St enables controlled small-batch processing and method optimization. For industrial manufacturing, sonicators equipped with flow-through reactors support continuous operation, high power density, and linear scale-up. These characteristics align with the requirements of pharmaceutical production environments, including process control and reproducibility.
In practical development workflows, Hielscher Ultrasonics systems are frequently used as reference platforms for ultrasonic liposome processing. At laboratory and formulation-development scale, compact ultrasonic probes such as the UP200Ht and UP400St enables controlled small-batch processing and method optimization. For industrial manufacturing, sonicators equipped with flow-through reactors support continuous operation, high power density, and linear scale-up. These characteristics align with the requirements of pharmaceutical production environments, including process control and reproducibility.
Beyond Semaglutide: A Platform Perspective
While semaglutide serves as a highly relevant model compound, the implications of ultrasonic liposome encapsulation extend beyond a single API. The same process logic applies to other lipidated peptides, peptide conjugates, and emerging biologics. As peptide therapeutics expand across metabolic disease, oncology, and immunology, scalable encapsulation technologies are likely to become decisive factors in determining which delivery strategies can progress from concept to commercial reality.
A Shift Toward Process-Engineered Peptide Delivery
Ultrasound-encapsulated liposomal semaglutide illustrates a broader shift in pharmaceutical development: from formulation concepts driven primarily by biological rationale toward delivery systems grounded in process engineering and manufacturability. In a field where many oral peptide technologies fail during scale-up, ultrasonic liposome processing offers a comparatively direct and technically robust pathway from laboratory development to industrial production.
Literature / References
- M.E. Barbinta-Patrascu, N. Badea, M. Constantin, C. Ungureanu, C. Nichita, S.M. Iordache, A. Vlad, S. Antohe (2018): Bio-Activity of Organic/Inorganic Photo-Generated Composites in Bio-Inspired Systems. Romanian Journal of Physics 63, 702 (2018).
- Raquel Martínez-González, Joan Estelrich, Maria Antònia Busquets (2016): Liposomes Loaded with Hydrophobic Iron Oxide Nanoparticles: Suitable T2 Contrast Agents for MRI. International Journal of Molecular Science 2016.
- Shah Purvin, Parameswara Rao Vuddanda, Sanjay Kumar Singh, Achint Jain, and Sanjay Singh (2014): Pharmacokinetic and Tissue Distribution Study of Solid Lipid Nanoparticles of Zidov in Rats. Journal of Nanotechnology, Volume 2014.
- Harshita Krishnatreyya, Sanjay Dey, Paulami Pal, Pranab Jyoti Das, Vipin Kumar Sharma, Bhaskar Mazumder (2019): Piroxicam Loaded Solid Lipid Nanoparticles (SLNs): Potential for Topical Delivery. Indian Journal of Pharmaceutical Education and Research Vol 53, Issue 2, 2019. 82-92.
Frequently Asked Questions
What are GLP-1 Peptides?
GLP-1 peptides are incretin-mimetic peptide drugs that activate the glucagon-like peptide-1 receptor (GLP-1R), a key metabolic receptor involved in glucose-dependent insulin secretion, suppression of glucagon release, delayed gastric emptying, and appetite regulation. Clinically used GLP-1 peptides (such as semaglutide) are chemically modified to resist enzymatic degradation and to achieve prolonged circulation time compared with native GLP-1.
What is the Difference between Semaglutide and Tirzepatide?
Semaglutide is a single‐agonist peptide that selectively activates the glucagon-like peptide-1 receptor (GLP-1R), whereas tirzepatide is a dual agonist that activates both the GLP-1 receptor and the glucose-dependent insulinotropic polypeptide receptor (GIPR). Biochemically, tirzepatide is a larger and more complex peptide with sequence elements and receptor-binding domains optimized to engage two incretin receptors, while semaglutide is engineered specifically for high-affinity GLP-1R activation. Both peptides are chemically modified with lipid moieties to increase plasma protein binding and prolong systemic half-life, but tirzepatide’s dual-receptor activity results in broader metabolic signaling.
How are Semaglutide and Tirzepatide Bio-chemically Classified?
Semaglutide is classified biochemically as a long-acting, lipidated GLP-1 receptor agonist peptide. Tirzepatide is classified as a long-acting, lipidated dual incretin receptor agonist peptide, specifically a GLP-1R/GIPR co-agonist.

Hielscher Ultrasonics manufactures high-performance ultrasonic homogenizers from lab to industrial size.