Sono-Electrochemistry and its Advantages
Here you will find all you need to know about ultrasonic electrochemistry (sonoelectrochemistry): working principle, applications, advantages and sono-electrochemical equipment – all relevant information about sonoelectrochemistry on one page.
Why Applying Ultrasonics to Electrochemistry?
The combination of low-frequency, high-intensity ultrasound waves with electrochemical systems comes with manifold benefits, which improve the efficiency and conversion rate of electrochemical reactions.
The Working Principle of Ultrasonics
For high-performance ultrasonic processing, high-intensity, low-frequency ultrasound is generated by an ultrasound generator and transmitted via an ultrasonic probe (sonotrode) into a liquid. High-power ultrasound is considered ultrasound in the range of 16-30kHz. The ultrasound probe expands and contracts e.g., at 20kHz, thereby transmitting respectively 20,000 vibrations per second into the medium. When the ultrasonic waves travel through the liquid, alternating high-pressure (compression) / low-pressure (rarefaction or expansion) cycles create minute vacuum bubbles or cavities, which grow over several pressure cycles. During the compression phase of the liquid and bubbles, the pressure is positive, while the rarefaction phase produces a vacuum (negative pressure). During the compression-expansion cycles, the cavities in the liquid grow until they reach a size, at which they cannot absorb more energy. At this point, they implode violently. The implosion of those cavities results in various highly energetic effects, which are known as the phenomenon of acoustic / ultrasonic cavitation. Acoustic cavitation is characterized by manifold highly energetic effects, which impact liquids, solid/liquid systems as well as gas/liquid systems. The energy-dense zone or cavitational zone is known as so-called hot-spot zone, which is most energy-dense in the close vicinity of the ultrasonic probe and declines with increasing distance from the sonotrode. The main characteristics of ultrasonic cavitation include locally occurring very high temperatures and pressures and respective differentials, turbulences, and liquid streaming. During the implosion of ultrasonic cavities in ultrasonic hot-spots, temperatures of up to 5000 Kelvin, pressures of up to 200 atmospheres and liquid jets with up to 1000km/h can be measured. These outstanding energy-intense conditions contribute to sonomechanical and sonochemical effects that intensify electrochemical systems in various ways.

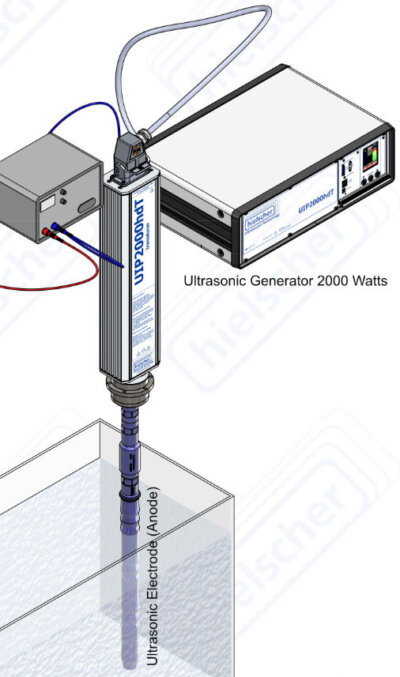
The probes of the ultrasonic processors UIP2000hdT (2000 watts, 20kHz) act as cathode and anode in an electrolytic cell
- Increases mass transfer
- Erosion / dispersions of solids (electrolytes)
- Disruption of solid/liquid boundaries
- High pressure cycles
The Effects of Ultrasonics on Electrochemical Systems
The application of ultrasonication to electrochemical reactions is known for various effects on the electrodes, i.e. anode and cathode, as well as the electrolytic solution. Ultrasonic cavitation and acoustic streaming generate significant micro-movement, impinging liquid jets and agitation into the reaction fluid. This results in improved hydrodynamics and movement of the liquid/solid mixture. Ultrasonic cavitation reduces the effective thickness of the diffusion layer at an electrode. A reduced diffusion layer means that sonication minimizes the concentration difference, meaning the convergence of the concentration in the vicinity of an electrode and the concentration value in the bulk solution are promoted ultrasonically. The influence of ultrasonic agitation on the concentration gradients during the reaction ensures the permanent feeding of fresh solution to the electrode and carting off of reacted material. This means sonication improved the overall kinetics accelerating reaction rate and increasing reaction yield.
By the introduction of ultrasonic energy into the system as well as the sonochemical formation of free radicals, electrochemical reaction, which otherwise would have been electroinactive, can be initiated.
Another important effect of acoustic vibration and streaming is the cleaning effect upon the electrode surfaces. Passivating layers and fouling at the electrodes limit the efficiency and reaction rate of electrochemical reactions. Ultrasonication keeps the electrodes permanently clean and fully active for reaction.
Ultrasonication is well-known for its degassing effects, which are beneficial in electrochemical reactions, too. Removing unwanted gases from the liquid, the reaction can run more efficacious.
- Increased electrochemical yields
- Enhanced electrochemical reaction speed
- Improved overall efficiency
- Reduced diffusion layers
- Improved mass transfer at the electrode
- Surface activation at the electrode
- Removal of passivating layers and fouling
- Reduced electrode overpotentials
- Efficient degassing of the solution
- Superior electroplating quality
Applications of Sonoelectrochemistry
Sonoelectrochemistry can be applied to various processes and in different industries. Very common applications of sonoelectrochemistry include the following:
- Nanoparticle synthesis (electrosynthesis)
- Hydrogen synthesis
- Electrocoagulation
- Wastewater treatment
- Breaking emulsions
- Electroplating / Electrodeposition
- Ohmic Heating
Sono-Electrochemical Synthesis of Nanoparticles
Ultrasonication was successful applied to synthesize various nanoparticles in an electrochemical system. Magnetite, cadmium-selenium (CdSe) nanotubes, platinum nanoparticles (NPs), gold NPs, metallic magnesium, bismuthene, nano-silver, ultra-fine copper, tungsten–cobalt (W–Co) alloy nanoparticles, samaria/reduced graphene oxide nanocomposite, sub-1nm poly(acrylic acid)-capped copper nanoparticles and many other nano-sized powders have been sucefully produced using sonoelectrochemistry.
Advantages of the sonoelectrochemical nanoparticle synthesis include the
- avoidance of reducing agents and surfactants
- use of water as a solvent
- adjustment of nanoparticle size by varying parameters (ultrasonic power, current density, deposition potential and the ultrasonic vs electrochemical pulse times)
Ashasssi-Sorkhabi and Bagheri (2014) synthesized polypyrrole films sonoelectrochemically and compared the results to electrocheically syntheised polypyrrole films. The results show that the galvanostatic sonoelectrodeposition produced a strongly adherent and smooth polypyrrole (PPy) film on steel, with a current density of 4 mA cm–2 in 0.1 M oxalic acid/0.1 M pyrrole solution. Using sonoelectrochemical polymerization, they obtained high-resistance and tough PPy films with smooth surface. It has been shown that PPy coatings prepared by sonoelectrochemistry provide substantial corrosion protection to St-12 steel. The synthesized coating was uniform and exhibited a high corrosion resistance. All these results may be attributed to the fact that the ultrasound enhanced the mass transfer of the reactants and caused high chemical reaction rates via acoustic cavitation and the resulting high temperatures and pressures. The validity of impedance data for the St-12 steel/two PPy coatings/corrosive media interface was checked using the KK transforms, and low average errors were observed.
Hass and Gedanken (2008) reported the successful sono-electrochemical synthesis of metallic magnesium nanoparticles. The efficiencies in the sonoelectrochemical process of the Gringard reagent in tetrahydrofuran (THF) or in a dibutyldiglyme solution were 41.35% and 33.08%, respectively. Adding AlCl3 to the Gringard solution increased the efficiency dramatically, raising it to 82.70% and 51.69% in THF or dibutyldiglyme, respectively.
Sono-Electrochemical Hydrogen Production
Ultrasonically promoted electrolysis significantly increases hydrogen yield from water or alkaline solutions. Click here to read more about the ultrasonically accelerated electrolytic hydrogen synthesis!
Ultrasonically Assisted Electrocoagulation
The application of low-frequency ultrasound to electrocoagulcation systems is known as sono-electrocoagulation. Studies show that sonication influences electrocoagulation positively resulting e.g., in higher removal efficiency of iron hydroxides from wastewater. The positive impact of ultrasonics on electrocoagulation is explained by the reduction of electrode passivation. Low-frequency, high-intensity ultrasound destructs deposited solid layer and removes them efficiently, thereby keeping the electrodes continuously fully active. Furthermore, ultrasonics activates both ion types, i.e. cations and anions, present in the electrodes reaction zone. Ultrasonic agitation results in high micro-movement of the solution feeding and carrying away raw material and product to and from the electrodes.
Examples for successful sono-electrocoagulation processes are the reduction of Cr(VI) to Cr(III) in the pharmaceutical wastewater, the removal of total phosphorus from effluents of the fine chemical industry with a phosphorous removal efficiency was 99.5% within 10 min., color and COD removal from effluents of the pulp and paper industry etc. Reported removal efficiencies for of color, COD, Cr(VI), Cu(II) and P were 100%, 95%, 100%, 97.3%, and 99.84%, respectively. (cf. Al-Qodah & Al-Shannag, 2018)
Sono-Electrochemical Degradation of Pollutants
Ultrasonically promoted electrochemical oxidation and/or reduction reactions are applied as powerful method to degrade chemical pollutant. Sonomechanical and sonochemical mechanisms promote the electrochemical degradation of pollutants. Ultrasonically generated cavitation results in intense agitation, micro-mixing, mass transfer and the removal of passivating layers from the electrodes. These cavitational effects result mainly in an enhancement of the solid-liquid mass transfer between the electrodes and the solution. Sonochemical effects directly impact molecules. Homolytic cleavage of molecules creates highly reactive oxidants. In aqueous media and in the presence of oxygen, radicals such as HO•, HO2• and O• are produced. •OH radicals are known to be important for the efficient decomposition of organic materials. Overall, sono-electrochemical degradation shows high efficiency and is suitable for the treatment of large volumes of wastewater streams and other polluted liquids.
For instance, Lllanos et al. (2016) found that significant synergistic effect were obtained for water disinfection when the electrochemical system was intensified by sonication (sono-electrochemical disinfection). This increase in the disinfection rate was found to be related to the suppression E. coli cell aggolomerates as well as an enhanced production of disinfectant species.
Esclapez et al. (2010) showed that a specifically designed sonoelectrochemical reactor (however not optimized) was used during the scale-up of trichloroacetic acid (TCAA) degradation, the presence of ultrasound field generated with the UIP1000hd provided better results (fractional conversion 97%, degradation efficiency 26%, selectivity 0.92 and current efficiency 8%) at lower ultrasonic intensities and volumetric flow. Considering the fact, that the pre-pilot sonoelectrochemical reactor was not yet optimized, it is very likely that these results can be even further improved.
Ultrasonic Voltammetry and Electrodeposition
Electrodeposition was carried out galvanostatically at current density of 15 mA/cm2. Solutions were subjected to ultrasonication before electrodeposition for 5–60 minutes. A Hielscher UP200S probe-type ultrasonicator was used at a cycle time of 0.5. Ultrasonication was achieved by directly dipping the ultrasound probe into the solution. To evaluate the ultrasonic impact on the solution before electrodeposition, cyclic voltammetry (CV) was used in order to reveal solution behavior and makes it possible to predict ideal conditions for electrodeposition. It is observed that when solution is subjected to ultrasonication before electrodeposition, deposition begins at less negative potential values. This means that at the same current in the solution less potential is required, as the species in the solution behave more active than in non-ultrasonicated ones. (cf. Yurdal & Karahan 2017)
Read more about sonoelectrochemical deposition of nano-enhanced coatings!
High-Performance Electrochemical Probes and SonoElectroReactors
Hielscher Ultrasonics is your long-time experienced partner for high-performance ultrasonic systems. We manufacture and distribute state-of-the-art ultrasonic probes and reactors, which are used worldwide for heavy-duty applications in demanding environments. For sonoelectrochemistry, Hielscher has developed special ultrasonic probes, which can act as cathode and/or anode, as well as ultrasonic reactor cells suitable for electrochemical reactions. Ultrasonic electrodes and cells are available for galvanic / voltaic as well as electrolytic systems.
Precisely Controllable Amplitudes for Optimum Results
 All Hielscher ultrasonic processors are precisely controllable and thereby reliable work horses in R&D and production. The amplitude is one of the crucial process parameters that influence the efficiency and effectiveness of sonochemically and sonomechanically induced reactions. All Hielscher Ultrasonics’ processors allow for the precise setting of the amplitude. Hielscher’s industrial ultrasonic processors can deliver very high amplitudes and deliver the required ultrasonic intensity for demanding sono-electrochamical applications. Amplitudes of up to 200µm can be easily continuously run in 24/7 operation.
All Hielscher ultrasonic processors are precisely controllable and thereby reliable work horses in R&D and production. The amplitude is one of the crucial process parameters that influence the efficiency and effectiveness of sonochemically and sonomechanically induced reactions. All Hielscher Ultrasonics’ processors allow for the precise setting of the amplitude. Hielscher’s industrial ultrasonic processors can deliver very high amplitudes and deliver the required ultrasonic intensity for demanding sono-electrochamical applications. Amplitudes of up to 200µm can be easily continuously run in 24/7 operation.
Precise amplitude settings and the permanent monitoring of the ultrasonic process parameters via smart software give you the possibility to influence the sonoelectrochemical reaction precisely. During every sonication run, all ultrasonic parameters are automatically recorded on a built-in SD-card, so that each run can be evaluated and controlled. Optimal sonication for most efficient sonoelectrochemical reactions!
All equipment is built for the 24/7/365 use under full load and its robustness and reliability make it the work horse in your electrochemical process. This makes Hielscher’s ultrasonic equipment a reliable work tool that fulfils your sonoelectrochemical process requirements.
Highest Quality – Designed and Manufactured in Germany
As a family-owned and family-run business, Hielscher prioritizes highest quality standards for its ultrasonic processors. All ultrasonicators are designed, manufactured and thoroughly tested in our headquarter in Teltow near Berlin, Germany. Robustness and reliability of Hielscher’s ultrasonic equipment make it a work horse in your production. 24/7 operation under full load and in demanding environments is a natural characteristic of Hielscher’s high-performance ultrasonic probes and reactors.
Contact us now and tell us about your electrochemical process requirements! We will recommend you the most suitable ultrasonic electrodes and reactor setup!
Contact Us! / Ask Us!
Literature / References
- Bruno G. Pollet; Faranak Foroughi; Alaa Y. Faid; David R. Emberson; Md.H. Islam (2020): Does power ultrasound (26 kHz) affect the hydrogen evolution reaction (HER) on Pt polycrystalline electrode in a mild acidic electrolyte? Ultrasonics Sonochemistry Vol. 69, December 2020.
- Md H. Islam; Odne S. Burheim; Bruno G.Pollet (2019): Sonochemical and sonoelectrochemical production of hydrogen. Ultrasonics Sonochemistry Vol. 51, March 2019. 533-555.
- Jayaraman Theerthagiri; Jagannathan Madhavan; Seung Jun Lee; Myong Yong Choi; Muthupandian Ashokkumar; Bruno G. Pollet (2020): Sonoelectrochemistry for energy and environmental applications. Ultrasonics Sonochemistry Vol. 63, 2020.
- Bruno G. Pollet (2019): Does power ultrasound affect heterogeneous electron transfer kinetics? Ultrasonics Sonochemistry Vol. 52, 2019. 6-12.
- Md Hujjatul Islam; Michael T.Y. Paul; Odne S. Burheim; Bruno G. Pollet (2019): Recent developments in the sonoelectrochemical synthesis of nanomaterials. Ultrasonics Sonochemistry Vol. 59, 2019.
- Sherif S. Rashwan, Ibrahim Dincer, Atef Mohany, Bruno G. Pollet (2019): The Sono-Hydro-Gen process (Ultrasound induced hydrogen production): Challenges and opportunities. International Journal of Hydrogen Energy, Volume 44, Issue 29, 2019, 14500-14526.
- Yurdal K.; Karahan İ.H. (2017): A Cyclic Voltammetry Study on Electrodeposition of Cu-Zn Alloy Films: Effect of Ultrasonication Time. Acta Physica Polonica Vol 132, 2017. 1087-1090.
- Mason, T.; Sáez Bernal, V. (2012): An Introduction to Sonoelectrochemistry In: Power Ultrasound in Electrochemistry: From Versatile Laboratory Tool to Engineering Solution, First Edition. Edited by Bruno G. Pollet. 2012 John Wiley & Sons, Ltd.
- Llanos, J.; Cotillas, S.; Cañizares, P.; Rodrigo, M. (2016): Conductive diamond sono-electrochemical disinfection 1 ( CDSED ) for municipal wastewater reclamation. Ultrasonics Sonochemistry Vol. 22, January 2015. 493-498.
- Haas, I.: Gedanken A. (2008): Synthesis of metallic magnesium nanoparticles by sonoelectrochemistry. Chemical Communications 15(15), 2008. 1795-1798.
- Ashassi-Sorkhabi, H.; Bagheri R. (2014): Sonoelectrochemical and Electrochemical Synthesis of Polypyrrole Films on St-12 Steel and Their Corrosion and Morphological Studies. Advances in Polymer Technology Vol. 33, Issue 3; 2014.
- Esclapez, M.D.; VSáez, V.; Milán-Yáñez, D.; Tudela, I.; Louisnard, O.; González-García, J. (2010): Sonoelectrochemical treatment of water polluted with trichloroacetic acid: From sonovoltammetry to pre-pilot plant scale. Ultrasonics Sonochemistry 17, 2010. 1010-1010.
- Sono-Electrochemical Synthesis Improves Efficiency in Chemical Manufacturing