Improved Fischer-Tropsch Catalysts with Sonication
Improved Synthesis of Fischer-Tropsch Catalysts with Ultrasound: Ultrasonic treatment of catalyst particles is used for several purposes. Ultrasonic synthesis helps to create modified or functionalized nano-particles, which have a high catalytic activity. Spent and poisoned catalysts can be easily and fast recovered by an ultrasonic surface treatment, which removes inactivating fouling from the catalyst. Finally, ultrasonic deagglomeration and dispersion results in a uniform, mono-disperse distribution of catalyst particles to ensure a high active particle surface and mass transfer for optimum catalytic conversion.
Advantages of Ultrasonic Catalyst Preparation for Fischer-Tropsch Processes
Sonication offers significant advantages in the synthesis of Fischer-Tropsch catalysts, primarily due to their ability to induce fine control over the catalyst morphology and active site distribution. The high-energy cavitation generated by ultrasonic waves ensures rapid mixing and the effective de-agglomeration of precursor materials, leading to a highly uniform particle size distribution and increased surface area. This enhanced homogeneity results in greater dispersion of active components, which is crucial for maximizing the number of accessible reaction sites. Furthermore, the controlled mixing kinetics often lead to the formation of highly stable and porous structures, thereby improving the catalytic performance, selectivity, and long-term stability of the catalyst under harsh reaction conditions.

Sonicator UIP1500hdT with flow-cell for the sonochemical synthesis of Fischer-Tropsch catalysts
Ultrasonic Effects on Catalysts
High power ultrasound is well-known for its positive influence on chemical reactions. When intense ultrasound waves are introduced into a liquid medium acoustic cavitation is generated. Ultrasonic cavitation produces locally extreme conditions with very high temperatures of up to 5,000K, pressures of approx. 2,000atm, and liquid jets of up to 280m/s velocity. The phenomenon of acoustic cavitation and its effects on chemical processes is known under the term sonochemistry.
A common application of ultrasonics is the preparation of heterogeneous catalysts: the ultrasound cavitation forces activate catalyst’s surface area as cavitational erosion generates unpassivated, highly reactive surfaces. Furthermore, mass transfer is significantly improved by the turbulent liquid streaming. The high particle collision caused by acoustic cavitation removes surface oxide coatings of powder particles resulting in the reactivation of the catalyst surface.
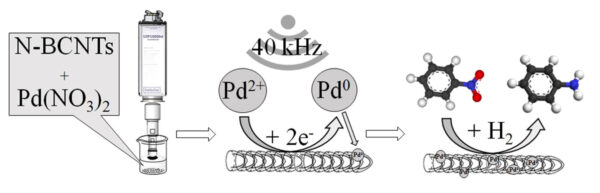
Synthesis of Palladium-doped catalyst using the sonicator UIP1000hdT
Study and image: ©Prekob et al., 2020
Ultrasonic Preparation of Fischer-Tropsch Catalysts
The Fischer-Tropsch process contains several chemical reactions that convert a mixture of carbon monoxide and hydrogen into liquid hydrocarbons. For Fischer-Tropsch synthesis, a variety of catalysts can be used, but most frequently used are the transition metals cobalt, iron, and ruthenium. The high temperature Fischer-Tropsch synthesis is operated with iron catalyst.
As Fischer-Tropsch catalysts are susceptible to catalyst poisoning by sulfur-containing compounds, the ultrasonic reactivation is of great importance to maintain full catalytic activity and selectivity.
- Precipitation or crystallization
- (Nano-) Particles with well-controlled size and shape
- Modified and functionalized surface properties
- Synthesis of doped or core-shell particles
- Mesoporous structuring
Ultrasonic Synthesis of Core-Shell Catalysts
Core–shell nanostructures are nanoparticles encapsulated and protected by an outer shell that isolates the nanoparticles and prevents their migration and coalescence during the catalytic reactions
Pirola et al. (2010) have prepared silica-supported iron-based Fischer-Tropsch catalysts with high loading of active metal. In their study is shown that the ultrasonically assisted impregnation of the silica support improves the metal deposition and increases the catalyst activity. The results of the Fischer-Tropsch synthesis have indicated the catalysts prepared by ultrasonication as the most efficient, particularly when ultrasonic impregnation is performed in argon atmosphere.

UIP2000hdT – 2kW powerful sonicator to prepare catalysts.
Ultrasonic Catalyst Reactivation
Ultrasonic particle surface treatment is a rapid and facile method to regenerate and reactivate spent and passivated catalysts. The regenerability of the catalyst allows for its reactivation and reuse and is thereby an economical and environmental-friendly process step.
Ultrasonic particle treatment removes inactivating passivating layers, fouling and impurities from the catalyst particle, which block sites for catalytic reaction. Sonicating a spent catalyst slurry results in jet washing the catalyst particle surface, thereby removing depositions from the catalytically active site. After ultrasonication, catalyst activity is restored to the same effectiveness as fresh catalyst. Furthermore, sonication breaks agglomerates and provides a homogeneous, uniform distribution of mono-dispersed particles, which increases the particle surface area and thereby the active catalytic site. Hence, ultrasonic catalyst recovery yields in regenerated catalysts with a high active surface area for improved mass transfer.
Ultrasonic catalyst regeneration works for mineral and metal particles, (meso-)porous particles and nanocomposites.
Read more about ultrasonic regeneration of spent catalysts!
High-Performance Sonicators for the Sonochemical Synthesis of Fischer-Tropsch Catalysts
 Hielscher sonicators are highly favored in catalyst synthesis due to their robust design, precision, and scalability, offering significant advantages over general sonication equipment. These units provide precisely controllable and high-intensity ultrasonic energy, which is critical for achieving uniform dispersion of precursor materials and facilitating precise nucleation and growth of catalyst particles. The sophisticated control systems allow researchers to accurately regulate parameters such as power output and pulse duration, ensuring reproducible experimental results–a vital factor in materials science. Furthermore, Hielscher sonicators are known for its durability and ability to handle various scales, ranging from small laboratory batches to pilot plant operations, thereby enabling the efficient transition of promising catalyst formulations from bench-scale research to industrial application. German engineering and manufacturing standards ensure that Hielscher ultrasonic equipment can be reliably operated in 24/7 operation under heavy-duty loads.
Hielscher sonicators are highly favored in catalyst synthesis due to their robust design, precision, and scalability, offering significant advantages over general sonication equipment. These units provide precisely controllable and high-intensity ultrasonic energy, which is critical for achieving uniform dispersion of precursor materials and facilitating precise nucleation and growth of catalyst particles. The sophisticated control systems allow researchers to accurately regulate parameters such as power output and pulse duration, ensuring reproducible experimental results–a vital factor in materials science. Furthermore, Hielscher sonicators are known for its durability and ability to handle various scales, ranging from small laboratory batches to pilot plant operations, thereby enabling the efficient transition of promising catalyst formulations from bench-scale research to industrial application. German engineering and manufacturing standards ensure that Hielscher ultrasonic equipment can be reliably operated in 24/7 operation under heavy-duty loads.
The table below gives you an indication of the approximate processing capacity of our sonicators:
| Batch Volume | Flow Rate | Recommended Devices |
|---|---|---|
| 1 to 500mL | 10 to 200mL/min | UP100H |
| 10 to 2000mL | 20 to 400mL/min | UP200Ht, UP400St |
| 0.1 to 20L | 0.2 to 4L/min | UIP2000hdT |
| 10 to 100L | 2 to 10L/min | UIP4000hdT |
| n.a. | 10 to 100L/min | UIP16000 |
| n.a. | larger | cluster of UIP16000 |
Contact Us! / Ask Us!
Facts Worth Knowing
What is the Fischer-Tropsch Reaction?
The Fischer-Tropsch reaction is a catalytic chemical process that converts synthesis gas, a mixture of carbon monoxide and hydrogen, into hydrocarbons such as alkanes, alkenes, waxes, and liquid fuels. It is an important route for producing synthetic fuels and chemicals from coal, natural gas, biomass, or CO₂-derived syngas.
What is a Fischer-Tropsch Catalyst?
A Fischer-Tropsch catalyst is a solid catalytic material that promotes the hydrogenation and chain-growth conversion of carbon monoxide with hydrogen into hydrocarbons. The most widely used active metals are iron, cobalt, and ruthenium, often supported on materials such as alumina, silica, titania, or carbon to improve surface area, stability, and selectivity.
What Industries use Fischer-Tropsch Reactions?
Fischer-Tropsch reactions are used in the synthetic fuels industry, petrochemical industry, gas-to-liquids production, coal-to-liquids production, biomass-to-liquids production, and emerging power-to-liquids and carbon capture utilization sectors. They are especially relevant for producing diesel, jet fuel, lubricants, waxes, olefins, and other hydrocarbon feedstocks.
What are Applications of Fischer-Tropsch Catalysts?
The Fischer–Tropsch synthesis is a category of catalytic processes that are be applied in the production of fuels and chemicals from synthesis gas (mixture of CO and H2), which can be
derived from natural gas, coal, or biomass the Fischer-Tropsch process, a transition metal-containing catalyst is used to produce hydrocarbons from the very basic starting materials hydrogen and carbon monoxide, which can be derived from various carbon-containing resources such as coal, natural gas, biomass, and even waste.
Literature / References
- Prekob, Á., Muránszky, G., Kocserha, I. et al. (2020): Sonochemical Deposition of Palladium Nanoparticles Onto the Surface of N-Doped Carbon Nanotubes: A Simplified One-Step Catalyst Production Method. Catalysis Letters 150, 2020. 505–513.
- Hajdu Viktória; Prekob Ádám; Muránszky Gábor; Kocserha István; Kónya Zoltán; Fiser Béla; Viskolcz Béla; Vanyorek László (2020): Catalytic activity of maghemite supported palladium catalyst in nitrobenzene hydrogenation. Reaction Kinetics, Mechanisms and Catalysis 2020.
- Pirola, C.; Bianchi, C.L.; Di Michele, A.; Diodati, P.; Boffito, D.; Ragaini, V. (2010): Ultrasound and microwave assisted synthesis of high loading Fe-supported Fischer–Tropsch catalysts. Ultrasonics Sonochemistry, Vol.17/3, 2010, 610-616.
- Suslick, K.S.; Hyeon, T.; Fang, M.; Cichowlas, A. A. (1995): Sonochemical synthesis of nanostructured catalysts. Materials Science and Engineering A204, 1995, 186-192.
Hielscher Ultrasonics manufactures high-performance ultrasonic homogenizers from lab to industrial size.


