Sonoelectrochemical Deposition
Sonoelectrochemical deposition is synthesis technique, which combines sonochemistry and electrochemistry, for a highly efficient and environmental-friendly production of nanomaterials. Renown as fast, simple, and effective, sonoelectrochemical deposition allows for shape-controlled synthesis of nanoparticles and nanocomposites.
Sono-Electrodeposition of Nanoparticles
For sonoelectrodeposition (also sonoeletrochemical deposition, sonochemical electroplating, or sonochemical electrodeposition) with the purpose of synthesizing nanoparticles, one or two ultrasonic probes (sonotrodes or horns) are used as electrodes. The method of sonoelectrochemical deposition is highly efficient as well as simple and safe to operate, which allows to synthesize nanoparticles and nanostructures in large quantities. Additionally, sonoelectrochemical deposition is an intensified process, meaning sonication accelerates the electrolysis process so that the reaction can be run under more efficacious conditions.
Applying power ultrasound to suspensions significantly increases mass transfer processes due to macroscopic streaming and microscopic interfacial cavitational forces. On ultrasonic electrodes (sono-electrodes), ultrasonic vibration and cavitation continuously removes the reaction products from the electrode surface. By removing any passivating depositions, the electrode surface is continuously available for new particle synthesis.
Ultrasound-generated cavitation promotes the formation of smooth and uniform nanoparticles which are homogeneously distributed in the liquid phase.
- Nanoparticles
- Core-Shell Nanoparticles
- Nanoparticle Decorated Support
- Nanostructures
- Nanocomposites
- Coatings
Sonoelectrochemical Deposition of Nanoparticles
 When an ultrasonic field is applied to a liquid electrolyte, diverse ultrasonic cavitation phenomena such as acoustic streaming and micro-jetting, shock waves, mass-transfer enhancement from/to the electrode and surface cleaning (removal of passivating layers) promote electrodeposition / electroplating processes. The beneficial effects of sonication on electrodeposition / electroplating already has been demonstrated for numerous nanoparticles, including metallic nanoparticles, semiconductor nanoparticle, core-shell nanoparticles and doped nanoparticles.
When an ultrasonic field is applied to a liquid electrolyte, diverse ultrasonic cavitation phenomena such as acoustic streaming and micro-jetting, shock waves, mass-transfer enhancement from/to the electrode and surface cleaning (removal of passivating layers) promote electrodeposition / electroplating processes. The beneficial effects of sonication on electrodeposition / electroplating already has been demonstrated for numerous nanoparticles, including metallic nanoparticles, semiconductor nanoparticle, core-shell nanoparticles and doped nanoparticles.
Sonochemically electrodeposited mettalic nanoparticles such as Cr, Cu and Fe show a significant increase in hardness, whilst Zn shows increased corrosion resistance.
Mastai et al. (1999) synthesized CdSe nanoparticles via sonoelectrochemical deposition. Adjustments of various electrodeposition and ultrasonic parameters allow to modify the crystal size of the CdSe nanoparticles from X-ray amorphous up to 9 nm (sphalerite phase).
Ashassi-Sorkhabi and Bagheri (2014) demonstrated the advantages of sono-electrochemical synthesis of polypyrrole (PPy) on St-12 steel in an oxalic acid medium using a galvanostatic technique with a current density of 4 mA/cm2. Direct application of low-frequency ultrasound using the ultrasonicator UP400S led to more compact and more homogeneous surface structures of polypyrrole. The results showed that the coating resistance (Rcoat), corrosion resistance (Rcorr), and Warburg resistance of ultrasonically prepared samples were higher than that of non-ultrasonically synthesized polypyrrole. Images of scanning electron microscopy visualized the positive effects of ultrasonication during electrodeposition on the particle morphology: The results reveal that the sonoelectrochemical synthesis yields strongly adherent and smooth coatings of polypyrrole. Comparing the results of sono-electro-deposition with conventional electrodeposition, it is clear that coatings prepared by the sonoelectrochemistry method have higher corrosion resistance. Sonication of the electrochemical cell results in enhanced mass transfer and in activation of the surface of the working electrode. These effects contribute significantly to a highly efficient, high-quality synthesis of polypyrrole.

Sonochemical electrodeposition allows to produce nanoparticles, core-shell nanoparticles, nanoparticle-coated support, and nanostructured materials.
(picture and study: ©Islam et al. 2019)
Sonoelectrochemical Deposition of Nanocomposites
The combination of ultrasonication with electrodeposition is efficacious and allows for a facile synthesis of nanocomposites.
Kharitonov et al. (2021) synthesized nanocomposite Cu–Sn–TiO2 coatings by sonochemical electrodeposition from an oxalic acid bath additionally containing 4 g/dm3 TiO2 under mechanical and ultrasonic agitation. Ultrasound treatment was performed with the Hielscher ultrasonicator UP200Ht at 26 kHz frequency and 32 W/dm3 power. Results demonstrated that ultrasonic agitation decreases agglomeration of TiO2 particles and allows for the deposition of dense Cu–Sn–TiO2 nanocomposites. When compared to conventional mechanical agitation, the Cu–Sn–TiO2 coatings deposited under sonication are characterized by higher homogeneity and smoother surface. In the sonicated nanocomposites, the majority of the TiO2 particles were embedded into the Cu–Sn matrix. The introduction of ultrasound agitation improves the surface distribution of the TiO2 nanoparticles and impedes aggregation.
It is shown that nanocomposite Cu–Sn–TiO2 coatings formed by ultrasonic-assisted electrodeposition exhibit excellent antimicrobial properties against E. coli bacteria.
High-Performance Sonoelectrochemical Equipment
Hielscher Ultrasonics supplies high-performance ultrasonic equipment for a reliable and efficient sono-electrodeposition / sonoelectroplating of nanomaterials. The product range includes high-power ultrasound systems, sono-electrodes, reactors and cells for your sono-electrochemical deposition application.
Contact Us! / Ask Us!
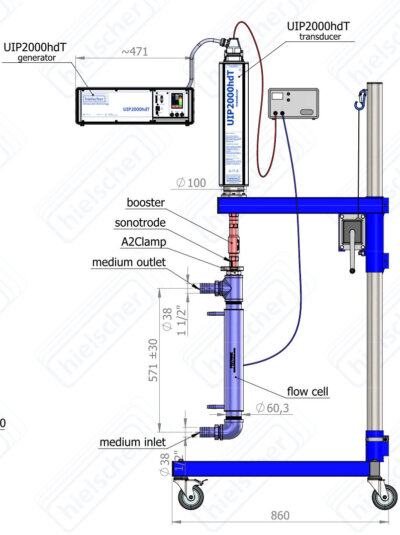 with ultrasonic probe UIP2000hdT for the electrodeposition of nanoparticles” width=”400″ height=”535″ class=”aligncenter size-P0400 wp-image-271130″ />
with ultrasonic probe UIP2000hdT for the electrodeposition of nanoparticles” width=”400″ height=”535″ class=”aligncenter size-P0400 wp-image-271130″ />
Literature / References
- Dmitry S. Kharitonov, Aliaksandr A. Kasach, Denis S. Sergievich, Angelika Wrzesińska, Izabela Bobowska, Kazimierz Darowicki, Artur Zielinski, Jacek Ryl, Irina I. Kurilo (2021): Ultrasonic-assisted electrodeposition of Cu-Sn-TiO2 nanocomposite coatings with enhanced antibacterial activity. Ultrasonics Sonochemistry, Volume 75, 2021.
- Ashassi-Sorkhabi, Habib; Bagheri, Robabeh (2014): Sonoelectrochemical and Electrochemical Synthesis of Polypyrrole Films on St-12 Steel and Their Corrosion and Morphological Studies. Advances in Polymer Technology 2014.
- Hyde, Michael; Compton, Richard (2002): How ultrasound influence the electrodeposition of metals. Journal of Electroanalytical Chemistry 531, 2002. 19-24.
- Mastai, Y., Polsky, R., Koltypin, Y., Gedanken, A., & Hodes, G. (1999): Pulsed Sonoelectrochemical Synthesis of Cadmium Selenide Nanoparticles. Journal of the American Chemical Society, 121(43), 1999. 10047–10052.
- Josiel Martins Costa, Ambrósio Florêncio de Almeida Neto (2020): Ultrasound-assisted electrodeposition and synthesis of alloys and composite materials: A review. Ultrasonics Sonochemistry, Volume 68, 2020.

Hielscher Ultrasonics manufactures high-performance ultrasonic homogenizers from lab to industrial size.





