Ekstrakcja białek wspomagana sonikacją dla fosfoproteomiki
In modern life sciences, phosphoproteomics has emerged as a cornerstone technology for decoding cellular signaling pathways and understanding disease mechanisms at a systems level. As phosphorylation governs critical biological functions – from enzyme activity to protein–protein interactions – its precise measurement is essential for both basic research and translational medicine. Recent advances in mass spectrometry have enabled the identification of tens of thousands of phosphorylation events in a single experiment, underscoring the need for robust, scalable, and reproducible sample preparation workflows.
Among the most impactful developments in this field is the adoption of sonication-assisted protein extraction, which significantly enhances sample quality, throughput, and reproducibility. Ultrasonic technologies such as the VialTweeter and the UIP400MTP are now redefining how laboratories handle large sample cohorts, particularly in high-throughput phosphoproteomics.
The Scientific Importance of Efficient Sample Preparation in Phosphoproteomics
 Protein phosphorylation is a highly dynamic and reversible post-translational modification affecting the majority of proteins in human cells. It regulates protein structure, localization, and interaction networks, and its dysregulation is implicated in diseases such as cancer and neurodegeneration.
Protein phosphorylation is a highly dynamic and reversible post-translational modification affecting the majority of proteins in human cells. It regulates protein structure, localization, and interaction networks, and its dysregulation is implicated in diseases such as cancer and neurodegeneration.
However, phosphoproteomic analysis presents unique technical challenges. Phosphorylated peptides are often low in abundance and require careful enrichment and highly efficient upstream sample preparation. Any inefficiency during protein extraction or digestion can lead to signal loss, poor reproducibility, and incomplete phosphosite coverage.
This is where sonication becomes critical.

Sonication of 40 autosampler vials with the UIP400MTP for protein extraction
Why Sonication is Transforming Protein Extraction
Sonication uses high-intensity ultrasound waves to mechanically disrupt cells and tissues, enabling efficient protein release from cells, tissues, biofluids and extracellular vesicles. Compared to conventional lysis techniques, sonication offers several distinct advantages:
- First, it ensures rapid and uniform cell disruption, which is particularly important for preserving transient phosphorylation states. In phosphoproteomics, even slight delays or incomplete lysis can alter signaling profiles, making fast and reproducible extraction essential.
- Second, sonication improves protein yield and solubilization, especially for difficult-to-lyse samples such as dense tissues or membrane-rich cells. This directly translates into better downstream digestion and phosphopeptide recovery.
- Third, ultrasonic processing is inherently scalable. Devices like the VialTweeter allow simultaneous sonication of multiple sealed tubes, ensuring identical processing conditions across samples. This eliminates variability introduced by manual handling.
- For even higher throughput demands, the UIP400MTP represents a major technological leap. It enables direct sonication of entire microplates or tube racks, including autosampler vials, making it ideal for processing hundreds of samples in parallel. This capability is particularly valuable in systems biology and clinical research, where large sample cohorts are standard.

VialTweeter: Simultaneous sonication of multiple samples under controlled process parameters
High-Throughput Sonication: VialTweeter and UIP400MTP in Focus
The integration of advanced ultrasonic devices into phosphoproteomic workflows is not merely a convenience – it is a methodological improvement.
The VialTweeter is designed for synchronized ultrasonic treatment of multiple closed vials, minimizing cross-contamination while ensuring reproducibility. It is especially suited for medium-throughput applications and standardized workflows.
In contrast, the UIP400MTP is optimized for high-throughput environments, enabling:
- Uniform sonication across entire standard microplates, e.g. 96-well or 384-well plates
- Direct processing of tube racks and autosampler vials
- Significant reduction in hands-on time
- Enhanced reproducibility across large datasets
This scalability aligns perfectly with modern phosphoproteomics approaches, where workflows routinely handle dozens to hundreds of samples in parallel.
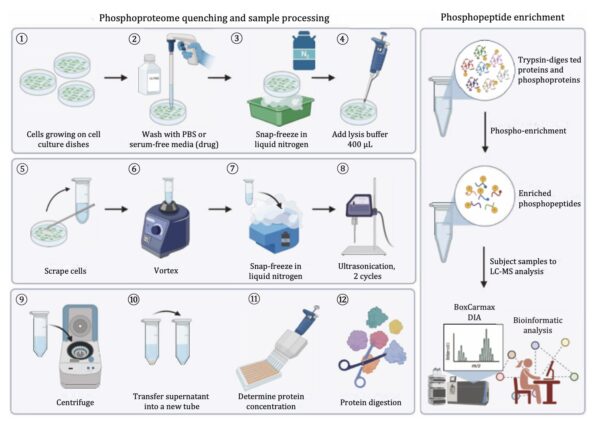
General Protocol for Sonication-Assisted Phosphoproteomic Sample Preparation
A robust phosphoproteomic workflow integrates efficient protein extraction, enzymatic digestion, and phosphopeptide enrichment. The following outline reflects established best practices adapted to sonication-based preparation:
- Rapid sample quenching and collection
Cells or tissues are rapidly quenched – commonly via snap-freezing – to preserve phosphorylation states. This step is crucial due to the transient nature of phosphorylation events. - Ultrasonic cell lysis and protein extraction
Samples are thawed and subjected to sonication, typically in short cycles. Ultrasonic energy disrupts cell membranes and releases proteins efficiently. In validated workflows, sonication is performed in multiple cycles to ensure complete lysis.
Example: After thawing the samples on ice, cell lysis was achieved by sonication, e.g., the Vialtweeter instrument for 2 cycles, each cycle 1 min. - Clarification and protein quantification
Following lysis, samples are centrifuged to remove debris. The supernatant containing soluble proteins is collected and quantified to ensure consistent input across samples. - Reduction and alkylation
Disulfide bonds are reduced (e.g., using DTT) and alkylated (e.g., using IAA) to stabilize proteins and improve digestion efficiency. - trawienie proteolityczne
Proteins are enzymatically digested, typically with trypsin, generating peptides suitable for mass spectrometry analysis. Read more about ultrasonically-accelerated protein digestion! - Peptide purification and desalting
Peptides are purified using C18-based methods to remove contaminants that could interfere with LC-MS analysis. - Phosphopeptide enrichment
Given the low abundance of phosphorylated peptides, enrichment techniques such as Fe-NTA or TiO₂ affinity methods are applied to selectively isolate phosphopeptides. - LC-MS/MS analysis and data processing
Enriched samples are analyzed using high-resolution mass spectrometry, often employing data-independent acquisition (DIA) for improved quantification and reproducibility.
Notably, large-scale workflows can be adapted to 96-well plate formats, enabling parallel processing of up to hundreds of samples – an approach fully compatible with UIP400MTP-based sonication.
Enhancing Reproducibility and Data Quality Through Sonication
One of the central challenges in phosphoproteomics is achieving consistent quantification across large datasets. Variability introduced during sample preparation can obscure biologically meaningful differences.
Sonication addresses this by providing:
- Standardized energy input across samples
- Reduced manual variability
- Improved reproducibility in protein extraction and digestion
When combined with high-throughput platforms like the UIP400MTP, laboratories can achieve a level of consistency that is essential for systems biology studies and clinical biomarker discovery.
The Future of Phosphoproteomics: Automation and Scalability
As phosphoproteomics continues to expand into large-scale and clinical applications, the demand for automation and throughput will only increase. Sonication-based sample preparation, particularly when integrated with microplate-compatible systems, represents a key enabling technology.
By combining efficient ultrasonic lysis, parallel processing, and compatibility with automated workflows, devices such as the VialTweeter and UIP400MTP are setting new standards in proteomics sample preparation.
Read more about the integration of the UIP400MTP in automated lab workflows!
Take Advantage of Sonication-Assisted Sample Prep in Phosphoproteomics!
Sonication-assisted protein extraction has become a critical component of modern phosphoproteomics, offering unmatched efficiency, scalability, and reproducibility. With the growing need to analyze complex biological systems across large sample cohorts, ultrasonic technologies are not just advantageous – they are essential.
By enabling high-throughput, standardized workflows, solutions like the VialTweeter and UIP400MTP are accelerating discoveries in cell signaling, disease mechanisms, and precision medicine.
Projektowanie, produkcja i doradztwo – Jakość Made in Germany
Ultradźwięki Hielscher są dobrze znane z najwyższej jakości i standardów projektowych. Solidność i łatwa obsługa pozwalają na płynną integrację naszych ultradźwiękowców z obiektami przemysłowymi. Trudne warunki i wymagające środowiska są łatwo obsługiwane przez ultradźwięki Hielscher.
Hielscher Ultrasonics jest firmą posiadającą certyfikat ISO i kładzie szczególny nacisk na wysokowydajne ultradźwięki z najnowocześniejszą technologią i łatwością obsługi. Oczywiście ultradźwięki Hielscher są zgodne z CE i spełniają wymagania UL, CSA i RoHs.
często zadawane pytania
What is Phosphoproteomics?
Phosphoproteomics is a specialized branch of proteomics that focuses on the large-scale identification, localization, and quantification of protein phosphorylation sites. It analyzes phosphorylated proteins and peptides to understand cellular signaling pathways, regulatory mechanisms, and dynamic post-translational modifications that control protein function.
Why is it Challenging to Ascertain Phosphorylation?
It is challenging to ascertain phosphorylation because phosphorylation events are often transient, low in abundance, and occur with low stoichiometry. Additionally, phosphorylated peptides can be difficult to detect due to their chemical instability, ionization variability in mass spectrometry, and the complexity of accurately assigning phosphorylation sites within peptide sequences.
What are Phosphoproteins?
Phosphoproteins are proteins that have undergone phosphorylation, meaning that one or more phosphate groups have been covalently attached to specific amino acid residues, typically serine, threonine, or tyrosine. This modification can alter the protein’s activity, structure, localization, or interactions with other molecules.
What is the Difference between Proteomics and Phosphoproteomics?
Proteomics is the comprehensive study of all proteins expressed in a biological system, including their abundance, structure, and interactions. Phosphoproteomics, in contrast, specifically focuses on the subset of proteins that are phosphorylated, emphasizing the analysis of phosphorylation sites and their regulatory roles in cellular processes.
Literatura / Referencje
- FactSheet UIP400MTP Plate-Sonicator for High-Throughput Sample Preparation – English version – Hielscher Ultrasonics
- FactSheet VialTweeter – Sonicator for Simultaneous Sample Preparation
- FactSheet UIP400MTP Plate-Sonicator für die High-Throughput Probenvorbereitung in 96-Well-Platten – deutsch – Hielscher Ultrasonics
- Di Y, Li W, Salovska B, Ba Q, Hu Z, Wang S, Liu Y. (2023): A basic phosphoproteomic-DIA workflow integrating precise quantification of phosphosites in systems biology. Biophysics Reports. 2023 Apr 30;9(2):82-98.
- Cosenza-Contreras M, Seredynska A, Vogele D, Pinter N, Brombacher E, Cueto RF, Dinh TJ, Bernhard P, Rogg M, Liu J, Willems P, Stael S, Huesgen PF, Kuehn EW, Kreutz C, Schell C, Schilling O. (2024): TermineR: Extracting information on endogenous proteolytic processing from shotgun proteomics data. Proteomics. 2024.
- Gao, Erli; Li, Wenxue; Wu, Chongde; Shao, Wenguang; di, Yi; Liu, Yansheng (2021): Data-independent Acquisition-based Proteome and Phosphoproteome Profiling across Six Melanoma Cell Lines Reveals Determinants of Proteotypes. Molecular Omics. 2021

Hielscher Ultrasonics produkuje wysokowydajne homogenizatory ultradźwiękowe od laboratorium do rozmiar przemysłowy.