Sonokemisk reduktion af palladium nanopartikler
Palladium (Pd) is well known for its catalytic properties and is also widely used in materials research, electronics manufacturing, medicine, hydrogen purification, and various chemical applications. Using a sonochemical route, the size and morphology of palladium particles can be controlled by adjusting the PVP/Pd ratio. This enables the ultrasonic synthesis of either very fine, monodisperse nanoparticles or larger palladium aggregates, allowing particle dimensions to be tailored for optimal catalytic performance.
Ultralydsproduktion af palladium nanopartikler
 Ultrasonic palladium nanoparticle reduction offers a fast, reagent-efficient route to Pd(0) nanoparticles by using acoustic cavitation to generate localized high-energy conditions and reducing radicals in solution, enabling palladium ions to be reduced without conventional high-temperature processing.
Ultrasonic palladium nanoparticle reduction offers a fast, reagent-efficient route to Pd(0) nanoparticles by using acoustic cavitation to generate localized high-energy conditions and reducing radicals in solution, enabling palladium ions to be reduced without conventional high-temperature processing.
A key advantage is process control: sonication time and stabilizer concentration, such as the PVP/Pd ratio, can influence whether the product forms as well-dispersed, rounded nanoparticles around 5 nm or as larger aggregates around 20 nm, which is industrially relevant because palladium performance in catalysis depends strongly on particle size, morphology, dispersion, and surface area. Since palladium nanoparticles are widely valuable as heterogeneous catalysts, electrocatalysts, and functional materials, ultrasonic reduction is attractive for producing finely dispersed Pd catalysts under comparatively mild liquid-phase conditions, with potential benefits for chemical synthesis, environmental catalysis, fuel-cell technologies, and other processes where high catalytic activity and efficient noble-metal utilization are economically important.

Industrial nanoparticle processing med sonikatoren UIP2000hdT
Sample Preparation Procedure
Prøverne blev fremstillet som følger:
Til prøverne, blandinger af 30 ml EG og 5,10-6mol of PVP were preprared by magnetic stirring for 15 min. For the different samples, different amount of Pd(NO₃)₂ solution, 1.5mL and 2mL, were added. The sample mixtures were prepared with the ratio of 2·10-3mol Pd(NO₃)₂ in sample (a) and 2.66·10-3mol Pd(NO₃)₂ in sample (b). Both mixtures were sonicated in a 20mL vial using a probe-type ultrasonicator. Samples were taken after sonication times of 30, 60, 90, 120, 150, and 180 min.
Analysen af forsøgsresultaterne viser, at:
- 1. Den sonokemiske reduktion af Pd (II) til Pd (0) afhænger af sonikeringstiden.
- 2. Det høje PVP/Pd(II) molforhold fører til dannelsen af monodispergerede palladiumpartikler med en afrundet form og en gennemsnitlig diameter på ca. 5nm.
- 3. Det lave PVP/Pd(II) molare forhold involverer imidlertid opnåelse af aggregater palladium nanopartikler med en stor størrelsesfordeling centreret ved 20nm.
Den sonokemiske vej til reduktion af palladium (II) ioner Pd(II) til palladiumatomer Pd(0) kan antages at være følgende:
- (1) Water pyrolysis: H₂O → •OH+•H
- (2) Radical formation: RH (Reducing agent) + •OH(•H) → •R + H₂O(H₂)
- (3) Ionreduktion: Pd(II) + reducerende radikaler (•H, •R) → Pd(0) + R•CHO + H+
- 4) Partikeldannelse: NPd(0) → Pdn
Resultat: Afhængigt af PVP/Pd(II)-forholdet, spredt eller aggregeret PdN blev opnået.
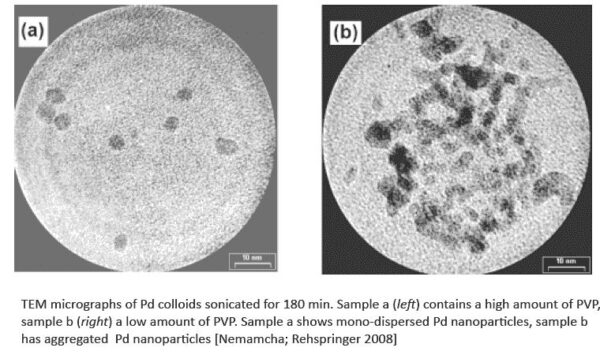
Sonochemical reduction of Palladium: sample a (left) contains a high amount of PVP, sample b (right) a low amount of PVP. Sonication time with UP100H: 180 min. Sample a shows mono dispersed Pd nano particles, sample b aggregated Pd nano particles.
Images and study: ©Nemamcha and Rehspringer, 2008
Analyse og resultater
De UV-synlige absorptionsanalyser bekræfter forholdet mellem den sonokemiske reduktion af palladium(II)-ioner til palladium(0)-atomer og retentionstiden i ultralydsfeltet. Reduktionen af palladium (II) ioner til palladium (0) atomer skrider frem og kan opnås fuldstændigt med stigende sonikeringstid. Mikrograferne af transmissionselektronmikroskopi (TEM) viser, at:
- When a high amount of PVP is added, the sonochemical reduction of palladium ions leads to the formation of monodispersed palladium particles with spherical shape and a mean diameter of approx. 5nm.
- The use of a small quantity of PVP involves the obtaining of aggregates palladium nanoparticles. The dynamic light scattering (DLS) measurements reveal that the palladium nanoparticles aggregates have a large size distribution centered at 20nm.

The lab sonicator UP100H has been used for the preparation of palladium nano-particles.
Design, produktion og rådgivning – Kvalitet fremstillet i Tyskland
Hielscher ultralydapparater er kendt for deres højeste kvalitet og designstandarder. Robusthed og nem betjening muliggør en jævn integration af vores ultralydapparater i industrielle faciliteter. Hårde forhold og krævende miljøer håndteres let af Hielscher ultralydsapparater.
Hielscher Ultrasonics er et ISO-certificeret firma og lægger særlig vægt på højtydende ultralydapparater med avanceret teknologi og brugervenlighed. Selvfølgelig er Hielscher ultralydapparater CE-kompatible og opfylder kravene i UL, CSA og RoHs.
Litteratur/Referencer
- Nemamcha, A.; Rehspringer, J. L. (2008): Morphology of dispersed and aggregated PVV-Pd nanoparticles prepared by ultrasonic irradiation of Pd(NO₃)₂ solution in ethylene glycol. Rev. Adv. Mater. Sci. 18;2008. 685-688.
- Prekob, Á., Muránszky, G., Kocserha, I. et al. (2020): Sonochemical Deposition of Palladium Nanoparticles Onto the Surface of N-Doped Carbon Nanotubes: A Simplified One-Step Catalyst Production Method. Catalysis Letters 150, 2020. 505–513.
- Haitao Zheng, Mphoma S. Matseke, Tshimangadzo S. Munonde (2019): The unique Pd@Pt/C core-shell nanoparticles as methanol-tolerant catalysts using sonochemical synthesis. Ultrasonics Sonochemistry, Volume 57, 2019. 166-171.
Fakta, der er værd at vide
What is Palladium?
Palladium is a rare, silvery-white precious metal with the chemical symbol Pd and atomic number 46. It belongs to the platinum-group metals and is valued because it is chemically stable, conducts electricity, absorbs hydrogen, and acts as an excellent catalyst. Finely divided palladium is especially effective for hydrogenation and dehydrogenation reactions, and heated palladium can allow hydrogen to diffuse through it, which makes it useful for hydrogen separation and purification.
What are Palladium Nanoparticles Used for?
Palladium nanoparticles are used mainly as high-surface-area catalysts. Because nanoparticles expose far more active surface area than bulk palladium, they can improve catalyst efficiency and reduce the amount of expensive noble metal required. Typical applications include chemical synthesis, hydrogenation reactions, carbon–carbon coupling reactions, electrocatalysis, fuel-cell research, hydrogen sensing and storage, environmental catalysis, and some biomedical research areas such as antimicrobial, photothermal, and anticancer systems. Palladium catalytic behavior depends strongly on particle size, morphology, and dispersion.
Palladium nanoparticles are also used to dope other particles in order to achieve catalytic functionalities. Read more about the ultrasonic pathway to synthesize Pd/N-BCNT as Fischer-Tropsch catalyst!
Is Palladium Toxic?
Elemental metallic palladium is generally considered to have low toxicity and no known biological role, but palladium compounds, salts, dusts, and nanoscale forms should be handled carefully. Occupational or laboratory exposure can cause irritation or sensitization depending on the compound and route of exposure, and palladium chloride solutions, for example, may irritate mucous membranes. For industrial handling, the practical answer is: bulk metallic palladium is relatively low-risk, but palladium powders, soluble palladium salts, and palladium nanoparticles should be treated as potentially hazardous materials, with dust control, ventilation, gloves, eye protection, and proper waste handling.

